第六章测试1.
The total spin of two electrons is ( ).
A:1 or 0 B:0
C:1
D:1/2
答案:A
2.
The maximum value of total angular momentum for electron with l=4 ( )
A:4.5
B:4 C:0 D:3.5 3.
The number of values allowed to total angular momentum j in Helium atom for a given non-zero l are ( )
A:3
B:0 C:1
D:2
4.
The 3s state of Na has an energy of -5.14 eV. Its effective nuclear charge is ( )
A:11e
B:1e
C:5e D:1.14 e 5.
The ground state of carbon is ( )
A:
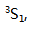
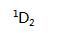
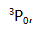
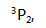
A system of electrons is described by a wave function should be ( )
A:symmetric
B:trial
C:anti-symmetric
D:no requirement 7.
The electron configuration of Neon is ( )
A:
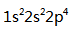
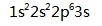
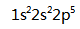
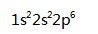

A:15 B:10 C:5 D:20 9.
What atoms have the configuration 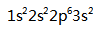 ( )
( )
A:Mg B: Si
C:Na D: Al 10.
How many electrons can be contained in M shell ( )
A:20
B:2
C:18
D:8 1.在正常塞曼效应中,沿磁场方向观察时将看到几条谱线( )
A:3
B:1
C:2
D:0
2.
下面哪一个是史特恩—盖拉赫实验证明正确的 ( )
A:角动量空间取向量子化,电子自旋假设是正确的,而且自旋量子数S=1/2
B:角动量空间取向量子化,但不能证明电子自旋假设正确的
C:其余选项都不对
3.在强外磁场中原子的附加能量ΔE除正比于B之外,同原子状态有关的因子有( )
A:磁量子数、朗德因子
B:朗德因子和玻尔磁子
C:

D:
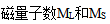
4.朗德因子反应了原子的内部运动。( )
A:对 B:错 5.对于很多元素,特别是较轻的元素来说,原子内部保持JJ耦合。( )
A:错 B:对 6.原子由于存在有效磁矩,在磁场当中会产生一部分附加能量。( )
A:对 B:错
温馨提示支付 ¥3.00 元后可查看付费内容,请先翻页预览!


